Knowing your soil’s pH is one of the keys to unlocking nutrient availability. By gaining insight on this common soil property measurement you can correct and prevent issues including nutrient deficiencies, diseases, and more.
Soil pH is a measurement of the alkalinity or acidity of soil. The degree of soil acidity measures the hydrogen ion concentration within the soil solution. Though changes in soil pH often occur slowly due to the buffering capacity of most mineral soils, knowing these changes can help you stay on top of any preventative or correctional measures needed.
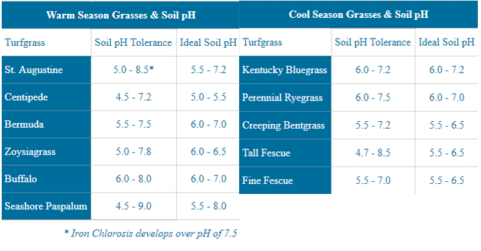
The pH scale ranges from 0-14, with levels above 7 to be considered alkaline and levels below to be considered acidic. Although a reading of 7 is known as neutral, the optimal pH range for turf species can generally fall anywhere between 5.0-7.0 (see below).
When pH levels are outside of that range it can adversely affect turfgrass growth and development, nutrient availability, pesticide effectiveness, weed reduction, and more.
Low pH
Acidic soil, or soil with a pH below 7, is created by the excess presence of hydrogen ions within the soil solution. This type of soil is commonly found along the East Coast, Pacific Northwest, and Southeastern areas of the United States. Low soil pH reduces microorganism activity, so thatch may accumulate more rapidly in an acidic soil. Soil with a pH of below 5 can be detrimental to turfgrass growth and can contribute to creating conditions favorable for pathogen activity.
High pH
Contrary to acidic soil, alkaline soil is created by the lack of hydrogen ions within the soil solution. Soil with a pH of greater than 7 is common in areas with a high clay content or in areas where the irrigation water is high in calcium or magnesium. Alkaline soils often lack availability to necessary nutrients, with iron, manganese, zinc, copper, and boron being the most prevalent. This decreased access to nutrients can result in deficiencies and diseases, such as Iron Chlorosis.
Other types of soil that are associated with a high pH reading are saline and sodic soils. Saline soils are high in calcium, magnesium or sodium salts and can adversely affect turfgrass quality. Sodic soils generally have a pH of over 8.5 and a high sodium content. This type of soil is often found in arid parts of the Western United States and lack good water infiltration due to the compaction of the soil. It is important to note that soils with a pH over 8 are often resistant to change pH levels because they are highly buffered.
Factors Affecting Soil pH
If your soil pH is outside of the recommended range for your turfgrass species, you might ask – How did this happen? What caused the pH to rise or fall? The answer is not so simple.
Since natural soil pH is a result of the combined effects of several soil-forming factors (parent material, timer, topography, climate, and the presence or absence of organisms), it’s hard to pinpoint exactly what caused a change. Broken down simply, there are three primary factors that affect soil pH: climate, mineral content, and soil texture.
Soil naturally loses some nutrients as water moves down through the soil in a process called leaching. The intensity of this process is primarily controlled by rainfall and temperature, with warm, humid environments being the most susceptible. When significant leaching is experienced due to heavy rainfall, soil pH decreases resulting in a process called soil acidification. On the flip side, dry climates experience soil weathering and leaching that is much less intense so the pH of soils in this climate are often neutral or alkaline.
Organic matter present within soil can also affect pH. For instance, soils with high clay and organic matter content are more resistant to fluctuations in pH levels (have a greater buffering capacity) than sandy soils. This difference in resistance is because sandy soils often have low organic matter content, which results in a low buffering capacity as well as high rates of water percolation and infiltration – making them more vulnerable to acidification.
How to Correct Soil pH
There are a few ways that soil pH can be raised or lowered, but due to the soils’ buffering capacity, doing so may differ in difficulty and is often temporary.
Raising Soil pH
- If soil pH is too low for optimal turf growth, then liming may be used to raise pH levels. Lime is calcium carbonate and is mined from quarries as stone and then crushed or granulated. The amount of lime needed depends on the soil pH, but generally, no more than 25-50 pounds per 1,000 ft2 per year is required to raise soil pH. Annual maintenance levels of lime are between 15-20 pounds per 1,000 ft2 per year.
Lowering Soil pH
- Sulfur or acidifying fertilizers like ammonium sulfate or iron sulfate may be used to lower the pH, but the change will be slow, and results are not often visible. The amount of sulfur will vary according to a soil test, but most turfgrasses can tolerate up to 5.0 pounds per 1,000 ft2.
Low or high pH can be corrected, but if caught too late, conditions may have already become favorable for disease, nutrient deficiencies, and other issues that are much harder to correct. Maintaining your soil’s pH within its target range can help to keep your turf healthy and thriving. To learn more about other factors contributing to your soil’s pH read our “Guide to Assessing and Managing Turfgrass Salinity Issues in Irrigation Water and Soils”.


Comments are closed.